|
In addition, the secondary endpoints did not meet statistical significance. The most common adverse experiences occurring with ACTIMMUNE therapy are “flu-like” symptoms such as fever, headache, chills, myalgia, or fatigue, which may decrease in severity as treatment continues, and may be minimized by bedtime administration of ACTIMMUNE.Horizon Pharma plc today announced that the Phase 3 trial, STEADFAST (Safety, Tolerability and Efficacy of ACTIMMUNE Dose Escalation in Friedreich's Ataxia study), evaluating ACTIMMUNE® (interferon gamma-1b) for the treatment of Friedreich's ataxia (FA) did not meet its primary endpoint of a statistically significant change from baseline in the modified Friedreich's Ataxia Rating Scale (FARS‐mNeuro) at 26 weeks versus treatment with placebo.At doses 10 times greater than the weekly recommended dose, ACTIMMUNE may exacerbate pre-existing cardiac conditions, or may cause reversible neurological effects such as decreased mental status, gait disturbance, and dizziness.Reversible neutropenia, thrombocytopenia, and elevations of AST and/or ALT have been observed during ACTIMMUNE therapy.Isolated cases of acute serious hypersensitivity reactions have been observed in patients receiving ACTIMMUNE.Acetaminophen may be used to prevent or partially alleviate the fever and headache The most common adverse experiences occurring with ACTIMMUNE therapy are “flu-like” symptoms such as fever, headache, chills, myalgia, or fatigue, which may decrease in severity as treatment continues, and may be minimized by bedtime administration of ACTIMMUNE.Avoid simultaneous administration of ACTIMMUNE with other heterologous serum protein or immunological preparations (eg, vaccines). 
Concomitant use of drugs with neurotoxic, hematotoxic, or cardiotoxic effects may increase the toxicity of interferons.Long-term effects of ACTIMMUNE on fertility are not known.ACTIMMUNE or nursing should be discontinued dependent on the importance of the drug to the mother Use of ACTIMMUNE by lactating mothers is not recommended. 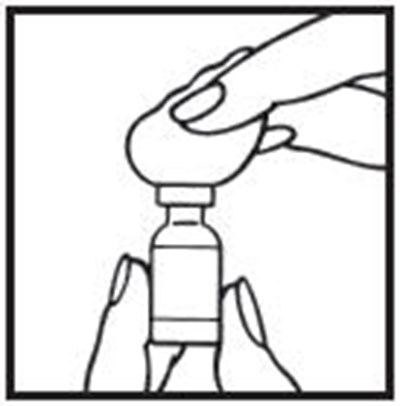
ACTIMMUNE should be used during pregnancy only if the potential benefit outweighs the potential risk to the fetus.Renal toxicity has been reported in patients receiving ACTIMMUNE Monitor renal function regularly when administering ACTIMMUNE in patients with severe renal insufficiency accumulation of interferon gamma-1b may occur with repeated administration.If severe hepatic enzyme elevations develop, ACTIMMUNE dosage should be modified Patients begun on ACTIMMUNE before age 1 year should receive monthly assessments of liver function.Myelosuppression, or receiving other potentially myelosuppressive agents consider dose reduction or discontinuation of therapy.Seizure disorders or compromised central nervous system function reduce dose or discontinue.Pre-existing cardiac conditions, including ischemia, congestive heart failure, or arrhythmia.ACTIMMUNE should be used with caution in patients with:.coli derived products, or any component of the product In patients who develop or have known hypersensitivity to interferon-gamma, E.IMPORTANT SAFETY INFORMATION CONTRAINDICATIONS 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
